
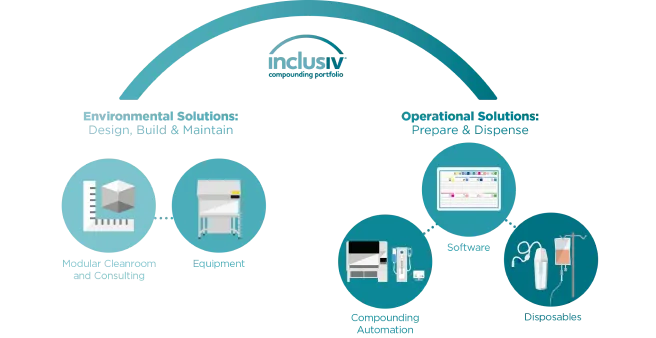
Modular Cleanroom & Consulting
Promote safety and compliance in the design, construction, and environmental control of the most stringent compounding settings.

Equipment
Enhance the safety and compliance of our cleanroom services and compounding devices with additional equipment.

Software
Efficiently manage your compounding devices and workflow by maintaining full visibility and traceability during each step of the compounding process.

Compounding Automation
Increase accuracy and control with automation that helps advance patient safety and protect staff during production of hazardous and non-hazardous compounded sterile preparations.
Disposables
Help improve patient safety and protect staff during the production and delivery of hazardous and non-hazardous compounded sterile preparations.
Need help with your sterile compounding plans?
Contact our experts to discuss your sterile compounding needs and future plans. Our team is here to support you.
Learn more about inclusiv Modular Cleanrooms
Want to receive product updates?
Sign up here to stay on top of the latest from inclusiv.
